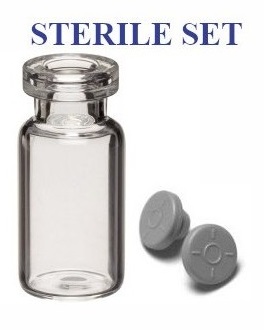
BUY Open Amber 30ml Sterile Vials: Ready to Use and Pyrogen-Free
Overview
Our Amber 30ml Sterile Vials are designed for the pharmaceutical manufacturing and compounding pharmacy sectors, particularly suited for cleanroom environments and Restricted Access Barrier Systems (RABS).
Product Details
- Construction Material: USP Type 1 Amber Borosilicate Glass
- Dimensions : 20mm crimp finish (13mm hole x 20mm outer rim), with a OD x H dimensions of 37mm (W) x 66mm (H)
- Packaging: Arranged in durable plastic trays of 63 vials, hermetically sealed for maximum protection.
Quality Assurance
Each vial is thoroughly processed and tested:
- Formed and washed with USP Water for Injection (WFI) by Gerresheimer®.
- Depyrogenated through heat treatment and sterilized using ethylene oxide by SteriGenics®.
- Comprehensive USP sterility and endotoxin testing reports are available for all lots, ensuring compliance with USP and cGMP regulations worldwide.
Additional Components Available
We also offer compatible 20mm seals and stoppers, available separately. Choose from washed, ready-to-sterilize options or pre-sterilized closures for your convenience.
Certification & Compliance of Closures
Our Ready to Use (RTU) vial stoppers and flip cap seals comply with stringent USP and AAMI standards. Our sterile vial closures are delivered ready to use, having undergone rigorous washing to eliminate particulates, inactivation of endotoxins, and sterilization according to USP and AAMI specifications. Third-party testing documentation by ISO certified testing laboratories is provided for all lots sold. This meticulous process reduces end-user costs and saves valuable time on processing, testing, and validation.
Why Choose Us?
We are your trusted source for complete vial packaging solutions tailored to meet the regulatory demands of the biological and pharmaceutical industries. Experience unparalleled quality and service with our products.
BUY 30ml amber open sterile vials online now – CLICK HERE
or
Worldwide delivery of 30ml amber open sterile vials from IVPACKS