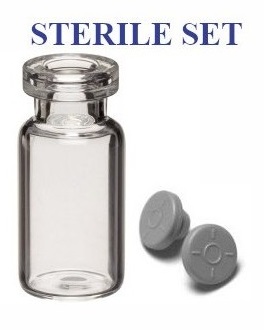
Sterile vial stoppers, washed and gamma irradiated, ready to use.
We offer 13mm and 20mm washed and sterilized vial stoppers for use with our open Ready to Fill sterile vials. Compounding pharmacies can save time and money while remaining in compliance with documentation requirements. Our sterile vial stoppers are manufactured from USP quality bromobutyl rubber, washed using purified water, packaged into 1,000 piece bags, and sent to be sterilized by gamma radiation. The manufacturers of our sterile vial stoppers are ISO certified. All production lots have 3rd party sterility and endotoxin testing performed with testing result documentation provided to our distributors and clients worldwide.